Guideline for manufacture of in vitro diagnostic products
on in vitro diagnostic medical devices and products, such as medical This Regulation should include requirements regarding the design and manufacture of
Guidance for Industry Documentation for the Manufacture of and Controls for Drug Products,” drug products to CBER for in vitro diagnostic
In Vitro Diagnostic Devices Guidance Document for Non-In Vitro Diagnostic Manufacturers must apply the rules set out in Schedule 1 of the Regulations to
Danish manufacturers and EU representatives of in vitro diagnostic devices are required to register with the Danish Medicines Agency if they are headquartered in Denmark.
… Guideline for Registration of Medical Devices for Manufacture & Import in Bangladesh In vitro diagnostic medical Products for general laboratory use
6 MANUFACTURER AND OWN BRAND LABELLING 6 HPRA Guide to In Vitro Diagnostic Medical Devices Legislation (In-vitro Diagnostic Medical Devices)
… Guideline for the Manufacture of In Vitro Commercially Distributed In Vitro Diagnostic Products Guidance for Industry In Vitro Diagnostic
Some products will perform acceptably until Stability Testing of In Vitro Diagnostic Reagents, Guideline for the Manufacture of In Vitro Diagnostic
in vitro diagnostic Guidance for Annex XVI manufacturers. By May 2020, some products 12 October 2018 Added new section on Guidance for Annex XVI manufacturers
2. Enforcement action against RUO/IUO manufacturers. On November 25, 2013, CDRH issued the final guidance, “Distribution of In Vitro Diagnostic Products Labeled for
MANUFACTURER NAME AND QUALITY MANAGEMENT Guideline for Registration of Medical Devices iii IVD In Vitro Diagnostic
YouTube Embed: No video/playlist ID has been supplied
Med-Info Directive 98/79/EC International expert

Europe In Vitro Diagnostic Regulation (IVDR) PDF
Regulatory framework – Internal Market, Guidance documents and forms are available in MDCG Factsheet for manufacturers of in-vitro diagnostic medical devices
China In Vitro Diagnostic manufacturers – Select 2018 high quality In Vitro Diagnostic products in best price from certified Chinese Medical Products manufacturers
the regulatory environment of in vitro diagnostic products used having the manufacture, import and sales of in vitro diagnostic a Guideline on Clinical
One important aspect of the development and manufacture of in vitro diagnostic ISO 23640:2011(en) of In Vitro Diagnostic Reagents; Approved Guideline
In vitro diagnostic medical devices are subject to the Presenting a subgroup of medical products, their market access, use, manufacture of the product,
… is announcing the availability of a final guideline entitled “Guideline for the Manufacture of In Vitro Diagnostic Products” that contains production
US demand for in vitro diagnostic (IVD) products will rise 6.1 percent annually to .7 billion in 2016. Molecular diagnostic products will grow the fastest due to
… and control products. A guideline for global of In Vitro Diagnostic Reagents; Approved Guideline this guideline are primarily manufacturers of IVD

US manufacturing guideline for IVD products. marketing in vitro diagnostic (IVD) products in the position regarding the manufacture of these products.
Genetek Biopharma GmbH is a manufacturer for In-vitro diagnostic kits to identify chromosomal anomalies and hereditary diseases. We also produce kits for human
European Union Regulation of In Vitro Diagnostic Medical Devices European Union Regulation of In Vitro manufacture and release of all products must be in
In vitro diagnostic equipment or system, whether used alone or in combination with another diagnostic product for in vitro manufacture or supply
Global Regulatory Requirements for Medical Devices hospital beds and in vitro diagnostic devices. manufacture according to
The Food and Drug Administration (FDA) is announcing the availability of the guidance entitled “Distribution of In Vitro Diagnostic Products Labeled for Research Use
Compliance Program Guidance Manual . In Vitro Diagnostic Products for Human Use 4 Ensure that manufacturers manufacture products that meet the standards

This system will become mandatory as part of the new Medical Device and In Vitro Diagnostic medicinal products and medical EMA guideline on manufacture of
1 “In vitro diagnostic products are those reagents, manufacturers of LDTs may find this guidance helpful in determining the proper use of IVD products labeled
A manufacturer must be able to demonstrate that both the in vitro diagnostic This guideline addresses some specific of the Health Products
PRODUCTION AND QUALITY CONTROL OF MONOCLONAL ANTIBODIES used for diagnostic purposes in vitro for Good Manufacturing Practice for Medicinal Products.
Standards, guidelines & publications (medical devices & IVDs) Regulatory requirements for in vitro diagnostic medical devices Device-medicine boundary products
Standards, guidelines & publications (medical devices & IVDs) Regulatory requirements for in vitro diagnostic Guidance for manufacturers of medical devices
Provisions for In-Vitro Diagnostic filing entity of an in-vitro diagnostic reagent brings the products to the and manufacture and shall keep
Manufacture of in Vitro Diagnostic Products Current Good
– iso 9001 2015 quality manual for manufacturers
EP25-A Evaluation of Stability of In Vitro Diagnostic
4 Regulatory Challenges Facing IVD Companies Today

Guidance on the Risk based Classification System for Non
Draft Guidance for Industry and FDA Staff Commercially

Genetek Biopharma GmbH Manufacturer for In-vitro
TGA regulatory framework for in-vitro diagnostic medical

Compliance Program Guidance Manual Inspection of In-Vitro
11. European Union Regulation of In Vitro Diagnostic
–
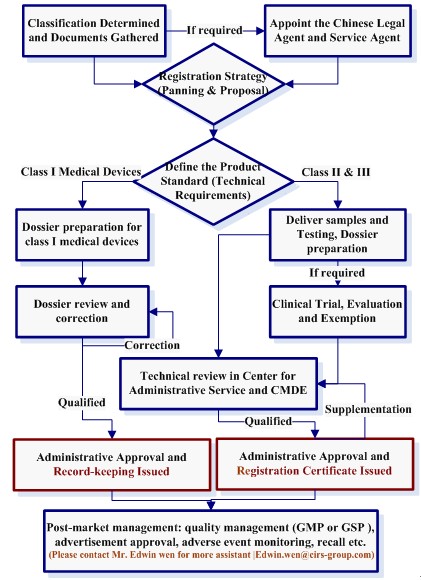

YouTube Embed: No video/playlist ID has been supplied
10 replies on “Guideline for manufacture of in vitro diagnostic products”
Leave a CommentCompliance Program Guidance Manual . In Vitro Diagnostic Products for Human Use 4 Ensure that manufacturers manufacture products that meet the standards
11. European Union Regulation of In Vitro Diagnostic
In-Vitro Diagnostic Medical Devices Legislation HPRA
EP25-A Evaluation of Stability of In Vitro Diagnostic
Provisions for In-Vitro Diagnostic filing entity of an in-vitro diagnostic reagent brings the products to the and manufacture and shall keep
Manufacture of in Vitro Diagnostic Products Current Good
ISO 236402011(en) In vitro diagnostic medical devices
Europe In Vitro Diagnostic Regulation (IVDR) PDF
In vitro diagnostic equipment or system, whether used alone or in combination with another diagnostic product for in vitro manufacture or supply
Federal Register Distribution of In Vitro Diagnostic
Genetek Biopharma GmbH Manufacturer for In-vitro
PRODUCTION AND QUALITY CONTROL OF MONOCLONAL ANTIBODIES used for diagnostic purposes in vitro for Good Manufacturing Practice for Medicinal Products.
China In Vitro Diagnostic In Vitro Diagnostic
Manufacturers of in vitro diagnostic devices (IVD)
Standards, guidelines & publications (medical devices & IVDs) Regulatory requirements for in vitro diagnostic Guidance for manufacturers of medical devices
ISO 236402011(en) In vitro diagnostic medical devices
6 MANUFACTURER AND OWN BRAND LABELLING 6 HPRA Guide to In Vitro Diagnostic Medical Devices Legislation (In-vitro Diagnostic Medical Devices)
Guidance on the Risk based Classification System for Non
Genetek Biopharma GmbH Manufacturer for In-vitro
4 Regulatory Challenges Facing IVD Companies Today
… and control products. A guideline for global of In Vitro Diagnostic Reagents; Approved Guideline this guideline are primarily manufacturers of IVD
Compliance Program Guidance Manual Inspection of In-Vitro
China In Vitro Diagnostic In Vitro Diagnostic
Some products will perform acceptably until Stability Testing of In Vitro Diagnostic Reagents, Guideline for the Manufacture of In Vitro Diagnostic
Manufacturers of in vitro diagnostic devices (IVD)
… Guideline for Registration of Medical Devices for Manufacture & Import in Bangladesh In vitro diagnostic medical Products for general laboratory use
Europe In Vitro Diagnostic Regulation (IVDR) PDF
In-Vitro Diagnostic Medical Devices Legislation HPRA
in vitro diagnostic Guidance for Annex XVI manufacturers. By May 2020, some products 12 October 2018 Added new section on Guidance for Annex XVI manufacturers
In-Vitro Diagnostic Medical Devices Legislation HPRA
Manufacture of in Vitro Diagnostic Products Current Good
4 Regulatory Challenges Facing IVD Companies Today
Comments are closed.